How Britain Lost Its (Dye) Empire
On the slow migration of an industry, and the forces that shaped its unlikely course
In the winter of 1916, as British soldiers headed to the Western Front during the First World War, they sang in jest:
We wore a tunic, a drab khaki tunic / You wore smart civvy clothes / We fought and fell at Loos while you were on the booze / That everybody knows.1
“Drab” as the khaki colored clothes may have been, they were nonetheless deeply desirable. Civilian military participation created a sense of patriotism around the war and the idea of the smartly dressed uniformed man became a symbol of pride.

Yet, despite how deeply entrenched the image of sharply cut servicemen was to become in the British milieu, the country struggled to dress its soldiers because it didn’t have enough synthetic dye to go around. In the sixty odd years preceding the war, Britain had been busy losing an industry it invented and should have dominated – a loss that eventually showed up in places stranger and graver than just clothing. The mechanics of the decline trace a mundane and unglamorous path involving university degrees, patent statutes, and hiring practices. However, the consequences were anything but trivial and ran from the color of a tunic to the chlorine gas at Ypres.
By the time WWI broke out in 1914, Germany controlled 85 percent of global dye production. That year, when Lord Kitchener needed to uniform half a million volunteers, the British army had to rely on bolts of dark blue serge instead – surplus fabric from the General Post Office.2 And so the men who had answered the most famous recruiting poster in history, marched off to war looking like postmen.

This was not without repercussions: “If army clothing was critical to how men performed their new military identities, then Kitchener blue played havoc with the physical and psychic transformation from civilian to soldier. Shapeless blue uniforms did not conform to the ideal image of the khaki service dress that was circulated in popular culture.”3 They also did not inspire confidence, after all a country that couldn’t color its own uniforms was now fighting one that had the systems in place to make anything it wanted out of coal tar – synthetic dye and poisonous gas.
The early beginnings
For all of recorded history, color had been an agricultural product with sources that were expensive, unreliable, and often unimaginably laborious to process. Indigo came from plantations in Bengal, madder from fields in Provence, cochineal from cactus farms in Mexico, Tyrian purple from sea snails dredged off the Levantine coast. Turkey red, the most prized cotton dye in Europe, took a process of nineteen separate steps spread over weeks. Yields (and prices) swung with the weather and, therefore, color was a luxury – the deeper or rarer the color, the more it cost.4
This changed in the spring of 1856 when William Henry Perkin, an 18-year-old chemistry student, attempted to synthesize quinine in a makeshift laboratory in his family’s East London flat. He failed, instead producing a reddish-brown sludge. However, rather than washing this concoction down the drain, Perkin decided to add alcohol to the residue. The solution turned a vivid purple.5
He had just invented mauve – the first synthetic dye. Within months, a Scottish dyer named Robert Pullar had written to him: “If your discovery does not make the goods too expensive it is decidedly one of the most valuable that has come out for a very long time, this color is one that has been very much wanted in all classes of goods.” Perkin, still a teenager, took out a patent, borrowed money from his father, and built a factory at Greenford Green.6
Now there was a color, a valuable one at that, which came from coal tar: the foul-smelling black waste product of gasworks, available by the ton in any industrial city.
Soon, Queen Victoria wore mauve velvet to the wedding of the Princess Royal and Punch magazine diagnosed a national epidemic of “mauve measles” a “form of mild insanity” that was “very catching.”7 By 1859 the color had conquered fashion from London to Paris, and Perkin, barely into his twenties, was rich.

The remarkable story of the color mauve is secondary at best, however, to what it proved: if you could make one dye from coal tar, you could theoretically make others. And within a decade, others did. Fuschine (soon renamed magenta) followed in Lyon in 1859, and by 1868 Graebe and Liebermann had synthesized alizarin, the principal coloring agent in madder root (a natural dye that had sustained major industries in France and the Netherlands for centuries).8
Germany takes notice
By 1900, synthetic dyestuffs had become an industry worth hundreds of millions of Francs (see figure below) and more than 1,200 distinct dyes were invented in the intervening decades9 – but who would make them, if not Britain?
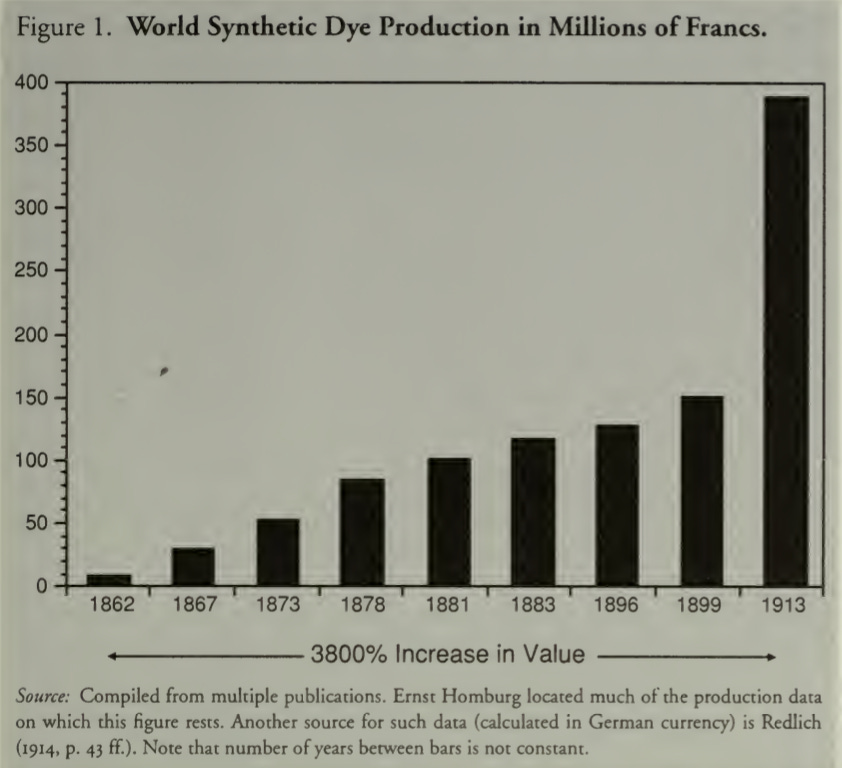
The answer, surprisingly and within a generation, was Germany (even though the synthetic dye industry was born in Britain) because in 1865, seven years after the initial discovery, August Wilhelm von Hofmann went back home to Berlin. He was a German chemist who had tutored Perkin at the Royal College of Chemistry, London and had seen this coming. As early as 1849, Hofmann had expressed “a hope that ere long we shall witness the production of the natural organic alkaloids and also of their allies the natural vegetable coloring matters artificially.”10
What followed was a transfer of industrial gravity so complete that it looks, in retrospect, like it was engineered. By 1870 Germany held about 50 percent of the global synthetic dye market. By 1913, Germany was producing roughly 85 percent of the world’s synthetic dyes while Britain managed 6.5 percent, France 5.4, the United States 3.3.11 These numbers become even more striking with the knowledge that Germany did not have cheaper raw materials, whereas Britain had more coal tar than it knew what to do with. Germany also did not have a large domestic market while both Britain and the United States had bigger textile industries.12
By 1900 German universities had roughly 2,200 chemistry students while British universities had a small fraction of that. The graduates of these institutions flooded into the dye firms: at Bayer, the chemist staff went from about 50 in 1890 to 262 by 1912.13 All of them held university doctorates; most had spent a year or two assisting one of the professors in Bayer’s academic network. Carl Duisberg, who built Bayer’s research operation and eventually ran the whole company, told an American audience in 1896 that “we choose almost exclusively young chemists from universities” and “always give preference to those, who after passing their examinations, have worked one or two years with one of the professors with whom we are associated.”14
Selected for their ability, these young chemists were productive. Bayer’s laboratories synthesized 2,656 new chemical compounds in 1906 alone of which sixty were tested at a larger scale and thirty-six reached the market. Six years earlier, Hoechst tested 3,500 new dyes (and dye combinations) and introduced eighteen.15 Such industry and invention can be ascribed to two reasons. For one, the firms were ruthlessly efficient and British chemist Henry E. Armstrong concluded upon visiting Bayer’s Elberfeld laboratory that:
“If, at the present time, it were desired to fit up a research laboratory for chemical purposes in London, we could not do better than take these plans [for the new Bayer laboratory] and reproduce them in their entirety, and that we should then, I believed, have reason to congratulate ourselves on possessing the best-appointed public research laboratory in the world.”16
Essentially, a private German corporate lab was the benchmark that a British public institution should aspire to.
The less magical part of the success was due to patent law. In Britain, inventors were allowed to patent chemical compounds themselves – Perkin patented mauve in 1856, and British law would let him do so for the dye itself. In contrast, Germany before 1877 had no effective patent regime at all, which meant any entrepreneur could enter the industry by copying innovations. When the early French and British patents expired, German firms could already produce the same dyes more cheaply. Thus, German start-ups proliferated in an environment of fierce competition.
After the unification of the country, lobbying began for a national patent law. However, the dye industry, which had everything to lose from a strict regime, won a crucial concession: the 1877 statute allowed only process patents for chemicals, pharmaceuticals, and food products. Industry participants understood by then that the absence of product protection had been crucial to their dominance in the global market – this was clearly worth preserving.17 For example, if one firm patented a route to alizarin, rivals could not copy it – but they could, and did, find another. Thus there was relentless incentive to innovate around existing processes. This led to tremendous competition and the firms guarded what they knew with something approaching paranoia. An employee at Bayer described the intense secrecy as such: “To keep the competition off our backs, we worked with rigged thermometers, every one showing 100 degrees too much,” so that anyone who bribed a worker for process data would get it wrong.18
Britain gives up on dye
Meanwhile, across the Channel, the British dye industry was dying. Perkin had retired in 1874, at thirty-six, a wealthy man in search of other scientific pursuits. He sold his operation to Brooke, Simpson & Spiller, once one of the largest British dye makers, who went bankrupt in 1905, and produced a rather telling postmortem: “The conditions for success [in the dye industry] are the employment of the best scientific knowledge, constant research and the adoption of modern and economical plant and process.” Arthur Green, writing during the war in 1917, looked back at the firm and noted that “no attempt was made by the firm to provide for the future by engaging more chemists.”19
The leading British survivor was Ivan Levinstein, who ran his Manchester firm like it was his personal fiefdom. Despite his rather crass management style, however, Levinstein understood perfectly well what was beating him. From his obituary: His name will always be remembered for his stout advocacy for the reform of the Patent Laws, which gave so unfair an advantage to the foreigner, and he undertook at great personal risk many successful actions against certain of the great German chemical firms in order to compel them to grant licences to manufacturers to work their patents in this country. As he once said, “they had patented the whole field of organic chemistry by their astute method of drafting their patents.” For his lobbying, meanwhile, the German firms punished Levinstein by refusing to buy the dozens of intermediate compounds (downstream of coal tar) that finished dyes required from his company.20
They were able to do this because “Germany makes all the dye-parts and the rest of the world merely assembles these dye-parts into finished dyes. Needless to say, the one who controls the manufacture of dye-parts actually controls the manufacture of dyes.” Such was the grim picture Bernhard Hesse, an American chemist, presented to the board of the General Chemical Company in New York in late 1914. The global coal-tar dye market consisted of about 900 finished dyes made from 300 intermediates derived from just nine coal-tar compounds. Of all these dyes available on the American market, only 100 were made or even assembled (from intermediates purchased from Germany) in the United States. Therefore, the transplanting of the industry out of Germany, he concluded, was “an undertaking properly and fitly to be described as titanic.”21
But people did take on the challenge. Victor Bloede, who co-founded American Aniline Works in West Virginia around 1878, recalled in a 1923 address how he and his partners had set out to break into the dye business. “Not one of us, of course, had any practical knowledge of any branch of this complicated art,” he admitted, “so we all began to read everything we could find on the subject of coal tar and its derivatives, including those wonderful German books of technology with titles something like ‘The Entire Art of Aniline Color Manufacturing in a Nutshell,’ ‘Every Man His Own Aniline Color Maker,’ ‘Simplified Manual of Producing Aniline Dyes,’ etc.”
After studying these useless texts the partners concluded that “the Germans were an utterly impractical people, and that by bringing genius into the business we could re-invent the art and reduce it to a much simpler and more practical form.” When this proved untrue, they advertised in German and English trade papers for a “correspondent” who could teach them by mail. The German trade paper that ran their advertisement was promptly raided by a state-backed bureau, the publisher was arrested, and Bloede and his partners were warned that any of them entering Germany would face a life sentence in a Zuchthaus (hard labor prison). American Aniline Works produced perhaps 20,000 pounds of fuchsine over its entire existence before the Little Kanawha River flooded in 1884 and tinted “the turbulent waters a beautiful raspberry shade for many miles” washed the plant into the Ohio, dyeing the river raspberry-red for miles.22
World War I and the dye famine
When WWI broke out in August 1914, the British naval blockade cut Germany off from its export markets almost overnight. Very quickly, by spring 1915, American newspapers were warning of a “dye famine” that threatened millions of workers in textiles, paint, paper, and printing.
This constraint on color was only the first major problem, the second was much worse. The same firms, chemists, and manufacturing plants working with the same intermediates that had built a formidable dye industry were now manufacturing explosives and poison gas.Carl Duisberg, then CEO of Bayer (who had told American audiences about his preference for young university chemists) whose laboratories had synthesized thousands of compounds in pursuit of better color, was a proponent for the military use of chemical agents. And so, the chlorine gas released at Ypres on 22 April 1915 was supplied by the German dye industry.

The men it killed included some of those volunteers who had marched to France in Post Office blue, those wearing the wrong color of uniform because Britain could not make its own dyes, were now breathing the wrong gas because Germany could turn its chemical industry to anything the army wanted.
Even then, the lesson wasn’t fully clear. As H. W. Richardson, writing in 1962, recalled – eighteen months after the outbreak of the war, the British government “was advertising for chemists of university training at a pay of 45 shillings per week; little more than an unskilled worker’s wage.”23
America joins the war – and the dye race
Things did change eventually though and under the Trading with the Enemy Act of October 1917, the United States government seized 4,500 German-owned dye and chemical patents – they paid a measly total compensation of $271,850.24 Woodrow Wilson also created the Chemical Foundation by executive order which was a non-profit corporation with ownership of the entire haul and they had a mandate to license the patents “upon equal terms and a royalty basis, to any bona fide American individual or corporation.”25
The largest American firm to take up the challenge was Du Pont. As Charles Reese, the company’s director of chemical research, explained to the National Research Council in 1921: “As we are manufacturers of explosives which require the same raw materials and similar processes to those used in the manufacture of dyes, we decided to branch out into the dye industry.”26 The qualification for making color was experience in making things blow up.
To start, they paid Ivan Levinstein (British dye mogul who worked to reform patent law in the country) £25,000 a year for ten years to hand over process secrets — including for synthetic indigo, which Levinstein had obtained from a former Hoechst plant confiscated by the British government. The Levinstein reports became “the bible” for Du Pont’s dye operation. In fact, so crucial were these reports that the researchers who carried them back across the Atlantic kept life preservers with them at all times, because Germany had vowed to “torpedo any neutral ship” (after Feb 3rd, 1917).
Even with such resources however, Du Pont could not make the dyes work. The problem, as its own historians later put it, “stemmed from the company’s trying to do in months what had taken the Germans six or seven decades to achieve.” The head of Du Pont’s technical laboratory, R. E. Rose admitted that “it takes nearly as much effort to decipher the correlation between patent and commercial dye as it does to discover the color originally.” The design of the German patent system, which protected processes, had made it so that the game-theory optimal move was to write patents in a way that was as unhelpful as possible.27 Therefore, it is of little surprise that Du Pont’s first attempts at indigo – the most important dye in the world – turned out green.
The Germans had a word for what Du Pont lacked: wissenschaftliche Massenarbeit, scientific mass work or the systematic, brute-force, industrialized method of invention that had produced thousands of new compounds in a single year at Bayer. Two of Du Pont’s chemists would later say: “No matter how much we may dislike to be followers and not pioneers, we must, in the first few years, confine our efforts in this field largely to the manufacture of colors that have already been produced by foreign manufacturers.”
In the end, Du Pont had to bring in these foreign manufacturers. Eric Kunz was dispatched to Lucerne to interview German chemists in hotel rooms, all of whom were offered $25,000 a year for five years, perhaps ten to fifteen times what these men were earning in Germany. They had, however, signed non-compete agreements (Karenz-Verpflichtungen) with their former employers. Two of the recruits were arrested at the Dutch border carrying a suitcase full of papers while two others made it to New York and were greeted on the dock with warrants for their arrest.

But others got through. Harold Elley remembered that “they knew their processes backwards and forwards,” William Calcott noted that the Germans were able “to assure the management that we were, after all, fundamentally on the right track.” Tacit knowledge of the German scientists helped Du Pont start seeing returns on their investment in the dye industry. Unsurprisingly there was a fair amount of criticism that the company faced to which Irénée du Pont replied: “Neither Germany nor the United States has a monopoly in brains, but there is a grave economic waste, both in money and time, in slowly and laboriously performing over again experiments which have already been made.”28
Du Pont’s total investment in dyestuffs eventually reached $40 million and American dye production surged from 6.6 million pounds in 1914 to 88 million by 1920.29
Britain too did not fight the entire war in postman blue. By May 1915, Levinstein Ltd had provided enough khaki dye to color the uniforms of nine million soldiers. The government created British Dyes Ltd in 1915 with public capital, and by 1926 the firm’s successor had been folded into Imperial Chemical Industries. A sixty year lag and one World War later, Britain finally had a chemical industry.
By tracing the ebbs and flows of the global distribution of the synthetic dye industry, it becomes clear that scientific breakthroughs require good institutions to flourish. Countries that invest in the infrastructure (in producing a conducive and stable research environment) tend to inherit the applied benefits of the discoveries.
Germany also did not out-compete Britain because German chemists were better. It did so because the German university system produced a larger volume of skilled graduates and because the patent structure rewarded firms that kept innovating rather than firms that had innovated once. Once these conditions were in place, an industrial cluster formed that was self-reinforcing: the universities trained chemists for the firms, the firms funded chairs at the universities,30 the patent system reduced gave firms enough protection to invest while keeping the field open enough to let knowledge compound
Eventually the dye industry found a new home again. The war made the transfer of technology necessary and the state made it possible by buying up patents and bringing on foreign talent. Discovery happens where it happens, but institutional capacity to absorb ideas and extend them is just as important. After all, process knowledge has spillovers that pure theory does not. The capacity to make dyes at scale turned out to be the capacity to make poison gas at scale.
With thanks to Mike Riggs, Stuart Buck, Elizabeth Van Nostrand, Jannik Reigl, Kevin Kohler, Abby ShalekBriski, and Venkatesh Ranjan for feedback and comments on drafts. Any errors within are mine alone.
A note: We recently launched the Research Revival Fund (grants for ideas that investigate lost or illegible research). Applications are now closed but if you are interested in being a patron or collaborator, please email me (hiya@mundane.beauty) or Wendi Yan (wendi@analoguegroup.org).
Harry Harding, From the City and the Plough: A Memoir of the 1914–18 War ,unpublished memoir, Department of Documents, IWM 80/28/1. As quoted in Jane Tynan, British Army Uniform and the First World War: Men in Khaki (Basingstoke: Palgrave Macmillan, 2013).
“The War Office did not obtain enough khaki uniforms in the opening weeks of the war, and many early recruits were forced to wear replacement uniforms, which became known as ‘Kitchener blue’. The uniforms were obtained from a variety of non-military sources: 500,000 suits of blue serge uniforms from post office stocks.” From Jane Tynan, British Army Uniform and the First World War: Men in Khaki (Basingstoke: Palgrave Macmillan, 2013), 47.
Ibid, 47-48.
“[At]no distant date. . . [England will be] the greatest color producing country in the world; nay, by the strangest revolutions, she may, ere long, send her coal-derived blues to indigo-growing India, her tar-distilled crimson to cochineal-producing Mexico and her fossil substitutes for quercitron and safflower to China andJapan, and the other countries whence these articles are now derived.” August Wilhelm von Hofmann, from ‘Report on the Chemical Section of the Exhibition of 1862.’ As quoted in Johann Peter Murmann, Knowledge and Competitive Advantage: The Coevolution of Firms, Technology, and National Institutions (Cambridge: Cambridge University Press, 2003), 32. More on natural dyes here: Wiki. Also see this wonderful essay on The Color of the Future by Étienne Fortier-Dubois
Anthony S. Travis, The Rainbow Makers: The Origins of the Synthetic Dyestuffs Industry in Western Europe (Bethlehem, PA: Lehigh University Press; London: Associated University Presses, 1993), 31-36.
Robert Pullar to William Henry Perkin, 12 June 1856, quoted in Murmann, Knowledge and Competitive Advantage, 109.
Jessica Durgan, “Color and Revolution in the Serialization of Dickens’s A Tale of Two Cities,” Victorians: A Journal of Culture and Literature 148 (2025).
C. J. Cooksey and A. T. Dronsfield, “Quirks of Dye Nomenclature. 4. Fuchsine: Four Shades of Magenta,” Biotechnic & Histochemistry 90, no. 4 (2015). And Carl Graebe and Carl Liebermann, “Ueber Alizarin und Anthracen,” Berichte der deutschen chemischen Gesellschaft 1 (1868): 49–51.
Eric Hagan and Jennifer Poulin, “Statistics of the Early Synthetic Dye Industry,” Heritage Science 9 (2021).
August Wilhelm von Hofmann, 1849, quoted in Murmann, Knowledge and Competitive Advantage, 6. Also see R. D. Welham, “The Early History of the Synthetic Dye Industry. I — The Chemical History,” Journal of the Society of Dyers and colorists 79, no. 3 (1963).
Johann Peter Murmann and Ernst Homburg, “Comparing Evolutionary Dynamics Across Different National Settings: The Case of the Synthetic Dye Industry, 1857–1914,” Journal of Evolutionary Economics 11, no. 2 (2001): 190-191, citing Thissen (1922, p. 18). The 85% figure for Germany’s market share is from Murmann, Knowledge and Competitive Advantage.
From Murmann and Homburg: “Why was the entry rate in the early years higher in Germany than in Britain even though Germany’s textile industry was about 23 times smaller than that of Britain?” and “The American textile market in 1913 was larger than that of France (approximately 3.5 times), Germany (2.5 times) and Switzerland (20 times).”
Lothar Burchardt, “Die Ausbildung des Chemikers im Kaiserreich,” Zeitschrift für Unternehmensgeschichte 23, no. 1 (1978): 31–53; Georg Meyer-Thurow, “The Industrialization of Invention: A Case Study from the German Chemical Industry,” Isis 73, no. 3 (1982): 363–381. See Figure 2 and Table 3.
“Once the student had reached a certain level of training, he would be assigned an experimental topic... After ten to twelve semesters he would complete his studies with the doctorate, though the dye-chemistry firms in particular desired that he then spend one to two years refining his laboratory skills as an assistant to ‘one of the professors with whom we are associated.’” C. Duisberg, Abhandlungen, Vorträge und Reden aus den Jahren 1882 bis 1921 (Berlin and Leipzig, 1923).
Bayer figures from Georg Meyer-Thurow, “The Industrialization of Invention.” 372 (Table 7); Hoechst figures from John J. Beer, The Emergence of the German Dye Industry (Urbana: University of Illinois Press, 1959), 89.
As quoted in Murmann, Knowledge and Competitive Advantage, 151.
Thomas Brenner and Johann Peter Murmann, “Using Simulation Experiments to Test Historical Explanations: The Development of the German Dye Industry 1857–1913,” Journal of Evolutionary Economics 26, no. 4 (October 2016): 911. And see “The practice of protection through secrecy that this encouraged came to an end with the passing of the 1877 Patent Law (the Reichspatentgesetz) that was uniform throughout Germany. This encouraged research and innovation through effective protection of new inventions. It later became a major incentive for the foundation of industrial research laboratories,” from Travis, The Rainbow Makers, 215.
Translation of Farbenfabriken-Bayer, 1918, p. 292 as seen in Murmann, Knowledge and Competitive Advantage, 137.
As quoted in Maurice R. Fox, Dye-Makers of Great Britain, 1856–1976: A History of Chemists, Companies, Products and Changes (Manchester: Imperial Chemical Industries PLC, 1987), 110; and Travis, The Rainbow Makers, 156 respectively.
For context: Britain had more coal tar than Germany, but coal tar is just the raw input. Turning it into the dozens of intermediate compounds that finished dyes required — aniline, anthraquinone, naphthalene derivatives — was a separate industrial operation. L. F. Haber, The Chemical Industry, 1900–1930: International Growth and Technological Change (Oxford: Clarendon Press, 1971), 148.
Bernhard C. Hesse, “The Industry of the Coal-Tar Dyes: An Outline Sketch,” Journal of Industrial and Engineering Chemistry 6, no. 12 (December 1914): 1013–1027.
Victor G. Bloede, “Some Early Attempts to Establish the Aniline Industry in the United States,” Industrial and Engineering Chemistry 16, no. 4 (April 1924)
As quoted in Murmann, Knowledge and Competitive Advantage, 155.
About $7 million today.
On the other side of the pond, under Annex VI of the Treaty of Versailles, Germany was required to hand over stocks of dyestuffs and to disclose its manufacturing processes to the Allies. Britain also created British Dyes Ltd. in 1915, which eventually became part of Imperial Chemical Industries in 1926.
David A. Hounshell and John Kenly Smith Jr., Science and Corporate Strategy: Du Pont R&D, 1902–1980 (Cambridge: Cambridge University Press, 1988), 79.
The “unclassified” dyes – the ones the Germans had deliberately kept off the patent rolls – had commanded a 40 to 50 percent price premium before the war, precisely because they revealed nothing about how they were made.
For this section: David A. Hounshell and John Kenly Smith Jr., Science and Corporate Strategy, 5 and 83-95.
Approximately more than $700 million; But when the postwar recession hit, Jackson Laboratory, at the heart of Du Pont’s dye research, went from 565 on the payroll to 217 in six months.
This was true at-least initially: “As German industrialization began to accelerate from mid-century, some sectors (notably synthetic dyestuffs) turned to the universities in their search for new products and manufacturing processes. Initially the cooperative arrangements between these firms and academic organic chemists were reasonably successful; the firms supplied reagents, suggested dissertation topics, engaged professors as consultants, and hired graduates.” And then took on a slightly different form towards the end of the 19th century, relying more heavily on the polytechnics and “ ‘third-sector institutions’ [that] were neither industrial nor academic in character. Their funding was usually ‘mixed’, coming from both public sources (including Reich, provincial, and/or local government) and private ones (wealthy individuals and/or firms).” From: Jonathan Harwood, “Institutional Innovation in Fin de Siècle Germany,” review of The Kaiser’s Chemists: Science and Modernization in Imperial Germany, by Jeffrey Allan Johnson, et al., British Journal for the History of Science 27, no. 2 (1994): 197–211.



